FOVISTA NR: Clinical Trial for neovascular age-related macular degeneration 30/01/2015
Description
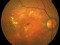
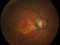
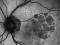
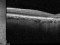
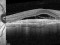
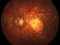
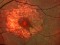
E10030-02 – An open-label investigator sponsored trial to investigate the safety, tolerability and development of subfoveal fibrosis by intravitreal administration of altering regimens of FOVISTATM (anti-PDGF-B PEGYLATED APTAMER) and anti-VEGF therapy (LUCENTIS) AVASTIN; or EYLEA) in subjects with neovascular age-related macular degeneration.
Key inclusion criteria
Subjects of either gender aged 50 years or above diagnosed with subfoveal choroidal neovascularization secondary to AMD.
Objectives
The objectives of the study are to evaluate the safety and efficacy of FOVISTA in combination with anti-VEGF THERAPY in patients with subfoveal nevascular age-related macular degeneration who have not responded to previous treatment.
If you are interested in taking part in a clinical trial, please fill in this form to contact us or call +34 935 950 155
Our team of professionals will answer you as soon as possible and we will assess whether you are eligible
Author
Dr Jordi Monés, médecin, titulaire d’un doctorat
Inscrit à l'ordre des médecins de Barcelone, sous le numéro 22838
Directeur
Docteur en médecine et chirurgie
Spécialiste en ophtalmologie
Spécialiste de la rétine, de la macula et du vitré