Institut de la Màcula, the first accredited european research site to treat atrophic AMD with Lampalizumab
02/02/2015 ·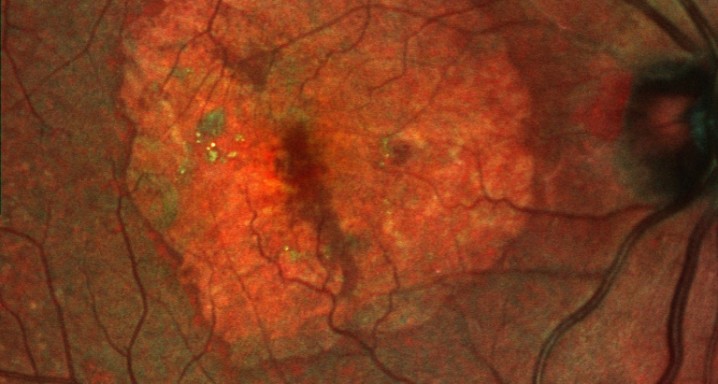
Lampalizumab is the fist therapy showing efficacy in significantly slowing down the progression of atrophic AMD, specially in patients with some genetic characteristics. Institut de la Màcula, part of the Hospital Quirón Teknon, the first accredited european research site to actively recruit patients in the first Phase III trial to treat and slow down the progression of the geographic atrophy secondary to AMD with Lampalizumab developed by Roche. Atrophic Age-related macular degeneration (AMD) is the main cause of non-preventable blindness in people of 50 years or older. This condition has no cure nowadays.
Phase III clinical studies have been initiated for lampalizumab, an investigational drug for geographic atrophy (GA), the advanced form of age-related macular degeneration (AMD), a progressive condition which can result in blindness. The phase III study program will evaluate the safety and efficacy of lampalizumab and its potential to slow the progression of GA. The studies will also further explore if people with a specific genetic biomarker, a mutation in complement factor I, may benefit more from lampalizumab treatment.
These phase III trials, called Chroma (GX29176) and Spectri (GX29185), are identically-designed, double-masked, randomized studies comparing 10 mg dose of lampalizumab administered every 4 or 6 weeks by intravitreal injection to sham injections. Approximately 936 patients will be enrolled in each study (188 biomarker-positive patients and 124 biomarker-negative patients each for the sham, lampalizumab q4w, and lampalizumab q6w treatment groups, in each study). The studies will recruit patients from approximately 300 physician-investigator sites in 24 countries worldwide. Key inclusion criteria are similar to that of the phase II MAHALO study and include the presence of geographic atrophy in both eyes with no history of neovascular (wet) AMD.
The primary objective of the studies is to demonstrate a reduction in the rate of GA disease progression. This efficacy endpoint, evaluated at one year (week 48), is defined as the mean change in the GA lesion area of the chosen study eye from baseline, as measured by fundus autofluorescence (FAF), an imaging technique used to provide information about the size and type of GA lesions in the macula. Secondary objectives of the studies, planned for evaluation at two years (96 weeks), focus on assessing the impact of lampalizumab treatment on patients’ visual function. If successful, long-term follow-up of patients completing Chroma and Spectri is planned to continue through a subsequent, open-label extension study.
The positive efficacy and safety results of previous Phase II trial, the MAHALO Study, supported this Phase II trials. The MAHALO study primary endpoint showed a 20 percent reduction in GA lesion progression in patients treated monthly with lampalizumab as compared with sham at month 18.1 Additionally, data from a sub-population of GA patients receiving monthly lampalizumab and positive for the complement factor I (CFI) biomarker, demonstrated a 44 percent decrease in the rate of disease progression at 18 months.
“Lampalizumab is a milestone in the journey to fight atrophic AMD. For the first time a drug has proven in a Phase II study efficacy in slowing down the progression of the deterioration. The Phase III is an opportunity for patients to have a chance to ameliorate the degeneration. The results of this Phase III trials may confirm these promising results” said Dr Jordi Monés, MD PhD and one of Principal Investigators in this Phase III Trial.