A clinical trial conducted by the Institut aims to show the efficacy and safety of Lampalizumab in intravitreal injections for the atrophic form of AMD
30/10/2015 ·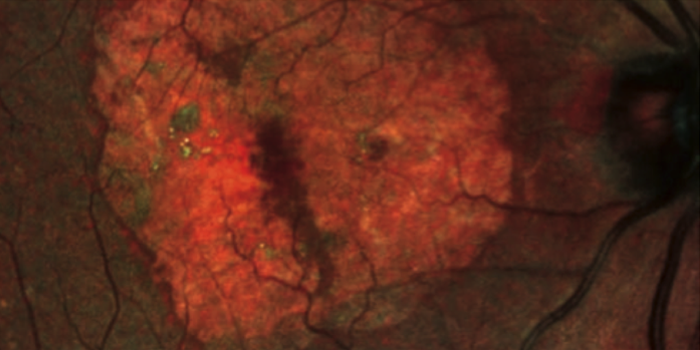
Lampalizumab is the first therapy to show significant efficacy in halting the progression of AMD in certain patients.
The research forms part of the programme of international clinical trials conducted by the Institut de la Màcula.
The trial aims to respond to the problems of patients with geographic atrophy (GA) secondary to Age-Related Macular Degeneration (AMD).
The Institut de la Màcula is the first centre in Europe to be accredited to conduct this trial.
Age-Related Macular Degeneration (AMD) in its dry or atrophic form is the primary cause of non-preventable blindness in the world in people of over 50 years old, especially from the age of 75. Lampalizumab is the first therapy to show significant efficacy in halting the progression of AMD, especially in patients with certain genetic characteristics.
The Institut de la Màcula, in one of the various benchmark international trials it conducts, assesses the efficacy and safety of Lampalizumab administered in the form of intravitreal injections to patients with geographic atrophy (GA) secondary to Age-Related Macular Degeneration (AMD). The Institut will also determine whether patients with a certain biomarker— a particular factor-1 mutation — may benefit more from treatment with Lampalizumab.
Geographic atrophy (GA) is a severe, advanced form of AMD that currently affects over 5 million people throughout the world. For those that suffer it, this pathology initially produces problems when reading, difficulty in recognising faces and problems in moving normally in poorly-lit spaces.
These phase III trials, technically called Chroma (GX29176) and Spectri (GX29185), are randomised, double-blind studies of identical design that compare a 10mg dose of Lampalizumab administered every 4 or 6 weeks through intravitreal injections to simulated ones. In each of these studies around 1,000 patients were recruited from approximately 300 research centres in 24 countries.
The principal inclusion criteria for these trials include the presence of geographic atrophy (GA) in both eyes of patients without a history of neovascular or wet AMD.
The main objective of the studies is to demonstrate that a reduction in the progression of geographic atrophy (GA) can been achieved. This efficacy assessment criterion, which will be evaluated after a year has passed, is defined as the mean variation in GA lesion area of the study eye, selected from baseline. It is measured by fundus autofluorescence (FAF), an imaging technique used to provide information on the extent and type of GA lesions in the macula.
The secondary aims of the studies, scheduled for evaluation after two years (96 weeks), focuses on assessment of the impact of treatment with Lampalizumab on patients’ visual function. If the studies are completed successfully, it is expected that there will be long-term monitoring of those patients to have completed Chroma and Spectri through a subsequent open extension study.
The positive efficacy and safety results of the previous Phase II trial, the MAHALO study, supports these Phase III trials. The principal assessment criterion of the MAHALO study showed a 20% fall after 18 months in the progression of the GA lesion in patients treated monthly with Lampalizumab as compared to the sham treatment.
In addition, the data from a subpopulation of GA patients who received Lampalizumab monthly and were positive for the complement factor I (CFI) biomarker, showed a 44% fall in the rate of progression of the disease after 18 months.
In the view of Dr. Jordi Monés: “Lampalizumab is a milestone in the fight against atrophic AMD. For the first time, a medicine has proven the efficacy of a Phase II study to reduce the progression of deterioration. Phase III is a chance for patients to halt their degeneration. The results of these Phase III trials can confirm these promising results”.
Lampalizumab, developed by Roche Pharma (Basle, Switzerland), is currently in the research phase to determine its effect on GA progression associated with advanced AMD. The Institut de la Màcula was the first centre to be accredited in Europe to conduct this trial.