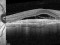
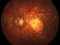
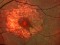
TREND: A phase IIIb, randomized, a 12-month, Visual Acuity assessor-masked, interventional study assessing the efficacy and safety of the Treat and Extend vs monthly regimens of 0.5 mg ranibizumab in patients with newly diagnosed nAMD 19/03/2014
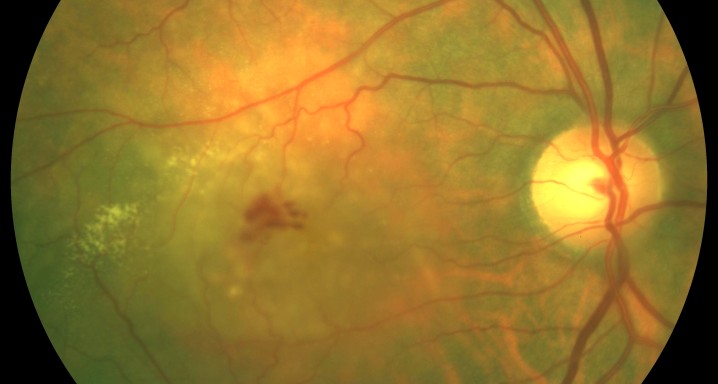
TREND Study: a 12-month, phase IIIb, randomized, Visual Acuity assessor-masked, multi-center, interventional study assessing the efficacy and safety of the Treat and Extend vs monthly regimens of 0.5 mg ranibizumab intravitreal (IVT) injections in patients with newly diagnosed nAMD. Patients will be randomized 1:1 into one of two treatment arms, Treat and Extend or monthly regimens.
Description
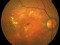
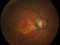
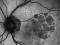
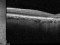
The Study aims to demonstrate that the efficacy and safety of ranibizumab (0.5mm) in a Treat-and-Extend protocol is no less than the monthly injection protocol for the treatment of age-related macular degeneration (AMD) of a wet or exudative type.
Up to now, the medicine Lucentis has been presented in a vial and has required filling. In this trial, for the first time in Spain, Lucentis is provided in a pre-filled syringe.
If you are interested in taking part in a clinical trial, please call us at 935 950 155 and we will assess whether you are eligible. Further information at clinicals trials.
Intravitreal injectionsExudative or wet AMDAuthor
Dr. Jordi Monés, M.D., Ph.D.
COMB Medical license number: 22.838
Director
Doctor of Medicine and Surgery
Specialist in Ophthalmology
Specialist in Retina, Macula and Vitreorretinal