Avonelle X: long-term safety and tolerability of the administration of intravitreal injections of Faricimab 04/12/2022
Description
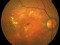
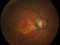
This study evaluates the long-term safety and tolerability of the administration of intravitreal injections of faricimab in patients with neovascular age-related macular degeneration (nAMD) that completed either of the two-phase III trials (GR40306 o GR40844). Additional exams include evaluation of efficacy, pharmacokinetics, immunogenicity and biomarkers.
Aim
The main objective is to evaluate the long-term ocular and systemic safety and tolerability of faricimab in patients with nAMD who have received at least one intravitreal injection during the previous trials, independently of treatment or protocol adherence.
Drug
Faricimab is the first bispecific drug for intraocular use that inhibits two mechanisms of action of the disease by blocking angiopoietin-2 (Ang-2) and vascular endothelial growth factor (VEGF-A).
Inclusion criteria
Only patients who have completed the GR40306 (TENAYA) or GR40844 (LUCERNE) trials without interruption in receiving the study drug can be enrolled in this study.
Duration
The duration of this study is two years.
Dry or atrophic AMD